Supply Chain
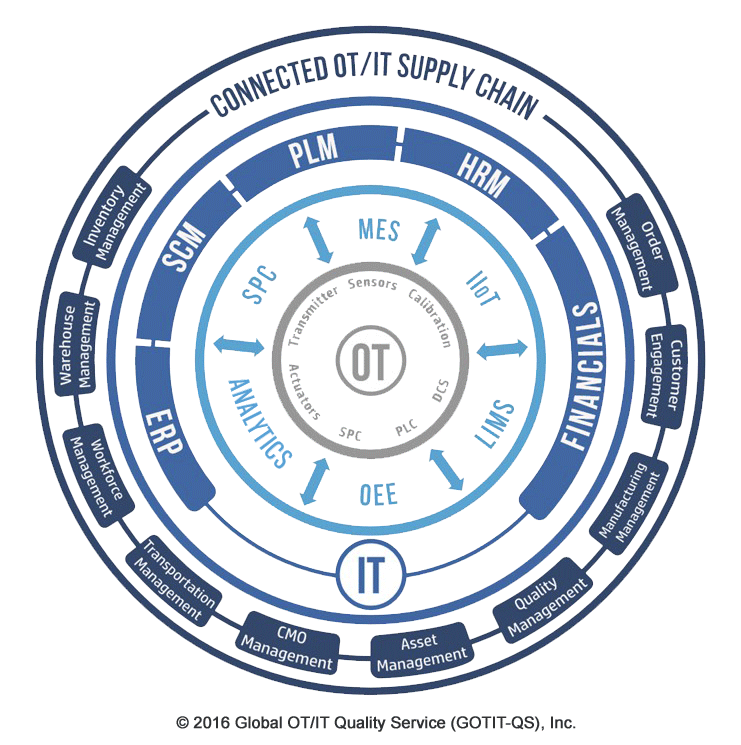
OT/IT Supply Chain
In the digitized manufacturing world represented above, the applications that support the business at all levels: from corporate to plant site, must be interacting by using compatible data models. In such a standardized environment, challenging business transformations including business acquisition or divesture; plant closing or introducing a new product become transparent and can approach the plug and play metaphore, or hitting the ground running.
To support The Connected Supply Chain, integration of business processes and data must exist at all layers including:
- The IT layer that includes the heavy duty business applications such as ERP, SCM, PLM, HRM and Financials
- The integration layer that provides the bridges between OT and IT that includes MES, SPC, Analytics, OEE, Lims and iiOT
- The OT layer that is the physical foundation for the entire edifice via Sensors, Actuators, Transmitters, Calibration, PLC, SPC and DCS elements
Global OT/IT Quality Services
Businesses evolve, reposition, and seek greater product market shares. Pharmaceuticals, Biotechnologies, and Chemical companies continue to acquire, divest, merge, and outsource manufacturing directly impacting and challenging the manufacturing supply chain while products move faster to market. The Automation / IT Application and Infrastructure landscape design and integration plays a key role and must be agile, scalable, and flexible enough to transforming and meeting changing business needs. Integrated IT architecture and proper selection of key manufacturing applications that meet business needs without impacting quality, speed to market, and interrupting the manufacturing supply chain is paramount. The advent of the Drug Quality and Safety Act (DQSA) is making the integration of OT and IT a central theme in any lifescience company. Serialization, aggregation and the dissemination of information throughout the supply chain is imperative for companies to continue to do business in the future. Anti-counterfeiting measures must be implemented to protect patients.
We refer to this OT/IT landscape and application integration as the “Connected Manufacturing Supply Chain.”
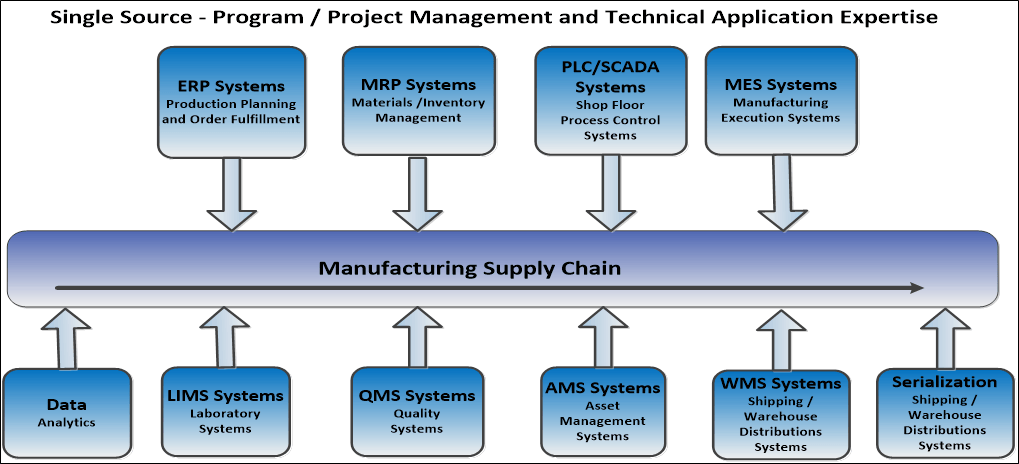
Single Source Program
Our extensive and proven services and solutions “plug-in” to enable businesses to function more effectively and directly impact "The Connected Supply Chain" to:
- Reduce time to market
- Reduce production cycle time
- Lower operational costs
- Increase collaboration with suppliers
- Accelerate warehouse throughput
- Optimize asset utilization